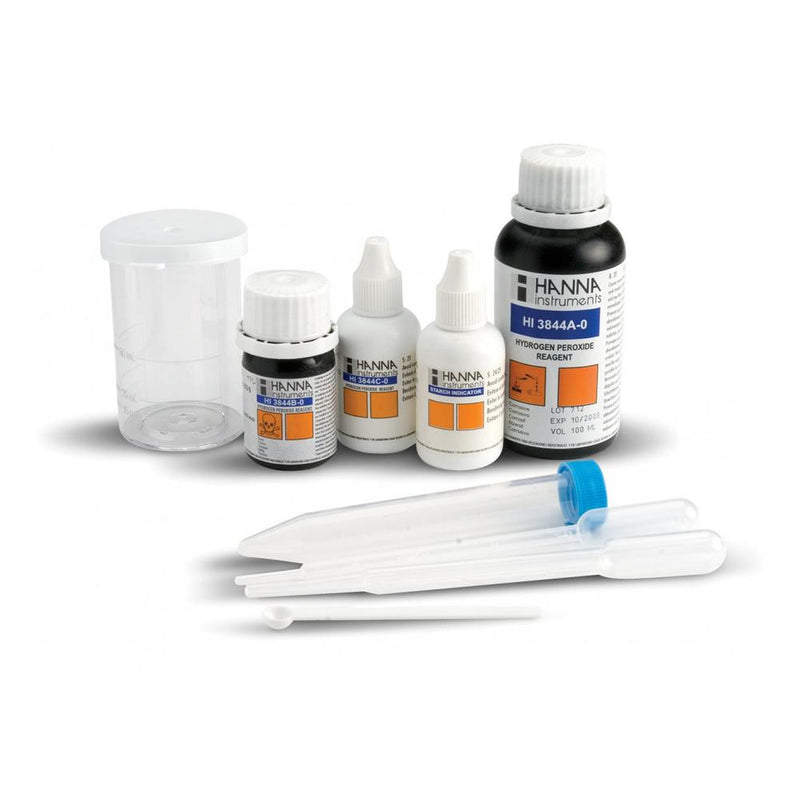
{"id":1582110341,"title":"HANNA HI3844 CTK Hydrogen Peroxide","handle":"hi3844","description":"\u003cp\u003eThe HI 3844 test kit can quickly and easy determine concentration in water up to \u003cbr\u003e 10 ppm of hydrogen peroxide. This is due to the fact that it is not affected by stabilizers. which are sometimes added to commercial hydrogen peroxide solutions. The kit is portable and can be used in the field as well as in the laboratory.\u003c\/p\u003e\n\u003cp\u003eHydrogen peroxide is determined by a titrimetric method. It reacts slowly with iodide in acid solution (Step 1); thus a 15 minute interval is required to allow the reaction to occur completely. The amount of iodine generated is equivalent to the hydrogen peroxide in the sample. The liberated iodine is then titrated with standard sodium thiosulfate solution that reduces the iodine back to iodide ions (Step 2).\u003c\/p\u003e\n\u003cp\u003eStep 1: H\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003eO\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003e + 2H\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e+\u003c\/span\u003e\u003c\/sup\u003e + 2I\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e—\u003c\/span\u003e\u003c\/sup\u003e → I\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003e + 2H\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003eO\u003cbr\u003e Step 2: I\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003e + 2(S\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003eO\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e3\u003c\/span\u003e\u003c\/sub\u003e)\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e2–\u003c\/span\u003e\u003c\/sup\u003e → 2I\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e—\u003c\/span\u003e\u003c\/sup\u003e + (S\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e4\u003c\/span\u003e\u003c\/sub\u003eO\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e6\u003c\/span\u003e\u003c\/sub\u003e)\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e2–\u003c\/span\u003e\u003c\/sup\u003e\u003c\/p\u003e\n\u003cp\u003e \u003c\/p\u003e\n\u003cp\u003e\u003cstrong\u003eOrder Information:\u003c\/strong\u003e\u003cbr\u003e HI 3844 test kit comes with 100 mL hydrogen peroxide reagent A. 17 g hydrogen peroxide reagent B. 30 mL hydrogen peroxide reagent C. 25 mL hydrogen peroxide reagent D. graduated plastic test tube with cap. 50 mL calibrated plastic vessel. 3 mL plastic pipette. 1 mL plastic pipette and plastic spoon.\u003c\/p\u003e\n\u003cdiv\u003e\u003cstrong\u003eProduct Manuals\u003c\/strong\u003e\u003c\/div\u003e\n\u003cp\u003e\u003cimg width=\"20\" height=\"22\" align=\"absmiddle\" alt=\"\" src=\"http:\/\/www.hannainst.com\/usa\/images\/icon-pdf.gif\" border=\"0\"\u003e Manual: \u003ca title=\"Download Product Manual\" href=\"http:\/\/www.hannainst.com\/manuals\/manHI_3844.pdf\" target=\"_blank\" rel=\"noopener noreferrer\"\u003eDownload\u003c\/a\u003e\u003c\/p\u003e\n\u003cp\u003e\u003cstrong\u003eSpecification :\u003c\/strong\u003e\u003c\/p\u003e\n\u003ctable class=\"spec_table\"\u003e\n\u003ctbody\u003e\n\u003ctr\u003e\n\u003ctd\u003eMethod\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003eTitration\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eRange\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003e0.00-2.00 mg\/L\u003cbr\u003e 0.0-10.0 mg\/L\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eSmallest Increment\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003e0.25 mg\/L\u003cbr\u003e 1.0 mg\/L\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eChemical Method\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003eIodometric\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eNumber of Tests\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003eapprox. 100\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eWeight\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003e450 g\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003c\/tbody\u003e\n\u003c\/table\u003e\n\u003cp\u003e \u003c\/p\u003e","published_at":"2017-11-22T12:52:58+13:00","created_at":"2015-09-01T15:08:58+12:00","vendor":"Hanna Instruments","type":"Electrochemical","tags":["Manufacturer_Hanna Instruments","Type_Chemical Test Kits"],"price":12200,"price_min":12200,"price_max":12200,"available":true,"price_varies":false,"compare_at_price":null,"compare_at_price_min":0,"compare_at_price_max":0,"compare_at_price_varies":false,"variants":[{"id":4857187013,"title":"Default Title","option1":"Default Title","option2":null,"option3":null,"sku":"HI3844","requires_shipping":false,"taxable":true,"featured_image":null,"available":true,"name":"HANNA HI3844 CTK Hydrogen Peroxide","public_title":null,"options":["Default Title"],"price":12200,"weight":0,"compare_at_price":null,"inventory_quantity":1,"inventory_management":null,"inventory_policy":"deny","barcode":null,"requires_selling_plan":false,"selling_plan_allocations":[],"quantity_rule":{"min":1,"max":null,"increment":1}}],"images":["\/\/acornsci.co.nz\/cdn\/shop\/products\/31gluhj8kul__sl500_aa300_51e06be9e5965_2015.jpeg?v=1465536739"],"featured_image":"\/\/acornsci.co.nz\/cdn\/shop\/products\/31gluhj8kul__sl500_aa300_51e06be9e5965_2015.jpeg?v=1465536739","options":["Title"],"media":[{"alt":"HI 3844 CTK Hydrogen Peroxide - Acorn Scientific","id":21281276003,"position":1,"preview_image":{"aspect_ratio":1.0,"height":1000,"width":1000,"src":"\/\/acornsci.co.nz\/cdn\/shop\/products\/31gluhj8kul__sl500_aa300_51e06be9e5965_2015.jpeg?v=1465536739"},"aspect_ratio":1.0,"height":1000,"media_type":"image","src":"\/\/acornsci.co.nz\/cdn\/shop\/products\/31gluhj8kul__sl500_aa300_51e06be9e5965_2015.jpeg?v=1465536739","width":1000}],"requires_selling_plan":false,"selling_plan_groups":[],"content":"\u003cp\u003eThe HI 3844 test kit can quickly and easy determine concentration in water up to \u003cbr\u003e 10 ppm of hydrogen peroxide. This is due to the fact that it is not affected by stabilizers. which are sometimes added to commercial hydrogen peroxide solutions. The kit is portable and can be used in the field as well as in the laboratory.\u003c\/p\u003e\n\u003cp\u003eHydrogen peroxide is determined by a titrimetric method. It reacts slowly with iodide in acid solution (Step 1); thus a 15 minute interval is required to allow the reaction to occur completely. The amount of iodine generated is equivalent to the hydrogen peroxide in the sample. The liberated iodine is then titrated with standard sodium thiosulfate solution that reduces the iodine back to iodide ions (Step 2).\u003c\/p\u003e\n\u003cp\u003eStep 1: H\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003eO\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003e + 2H\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e+\u003c\/span\u003e\u003c\/sup\u003e + 2I\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e—\u003c\/span\u003e\u003c\/sup\u003e → I\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003e + 2H\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003eO\u003cbr\u003e Step 2: I\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003e + 2(S\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e2\u003c\/span\u003e\u003c\/sub\u003eO\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e3\u003c\/span\u003e\u003c\/sub\u003e)\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e2–\u003c\/span\u003e\u003c\/sup\u003e → 2I\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e—\u003c\/span\u003e\u003c\/sup\u003e + (S\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e4\u003c\/span\u003e\u003c\/sub\u003eO\u003csub\u003e\u003cspan style=\"font-size: small;\"\u003e6\u003c\/span\u003e\u003c\/sub\u003e)\u003csup\u003e\u003cspan style=\"font-size: small;\"\u003e2–\u003c\/span\u003e\u003c\/sup\u003e\u003c\/p\u003e\n\u003cp\u003e \u003c\/p\u003e\n\u003cp\u003e\u003cstrong\u003eOrder Information:\u003c\/strong\u003e\u003cbr\u003e HI 3844 test kit comes with 100 mL hydrogen peroxide reagent A. 17 g hydrogen peroxide reagent B. 30 mL hydrogen peroxide reagent C. 25 mL hydrogen peroxide reagent D. graduated plastic test tube with cap. 50 mL calibrated plastic vessel. 3 mL plastic pipette. 1 mL plastic pipette and plastic spoon.\u003c\/p\u003e\n\u003cdiv\u003e\u003cstrong\u003eProduct Manuals\u003c\/strong\u003e\u003c\/div\u003e\n\u003cp\u003e\u003cimg width=\"20\" height=\"22\" align=\"absmiddle\" alt=\"\" src=\"http:\/\/www.hannainst.com\/usa\/images\/icon-pdf.gif\" border=\"0\"\u003e Manual: \u003ca title=\"Download Product Manual\" href=\"http:\/\/www.hannainst.com\/manuals\/manHI_3844.pdf\" target=\"_blank\" rel=\"noopener noreferrer\"\u003eDownload\u003c\/a\u003e\u003c\/p\u003e\n\u003cp\u003e\u003cstrong\u003eSpecification :\u003c\/strong\u003e\u003c\/p\u003e\n\u003ctable class=\"spec_table\"\u003e\n\u003ctbody\u003e\n\u003ctr\u003e\n\u003ctd\u003eMethod\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003eTitration\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eRange\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003e0.00-2.00 mg\/L\u003cbr\u003e 0.0-10.0 mg\/L\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eSmallest Increment\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003e0.25 mg\/L\u003cbr\u003e 1.0 mg\/L\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eChemical Method\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003eIodometric\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eNumber of Tests\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003eapprox. 100\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003ctr\u003e\n\u003ctd\u003eWeight\u003c\/td\u003e\n\u003ctd\u003e \u003c\/td\u003e\n\u003ctd\u003e450 g\u003c\/td\u003e\n\u003c\/tr\u003e\n\u003c\/tbody\u003e\n\u003c\/table\u003e\n\u003cp\u003e \u003c\/p\u003e"}
HANNA HI3844 CTK Hydrogen Peroxide
Related Products
Carbolite Heating element 200mm long
$799.00 $850.00
Carbolite Thermocouple for AAF11/3
$299.00 $464.00
Carbolite Thermocouple for AAF11/7
$299.00 $464.00
Carbolite Thermocouple for AAF11/7
$299.00 $464.00
HANNA BL983320-1 Conductivity Mini Controller @ 199.9 µS/cm
$350.00 $465.00